Why Soil Extractants Matter
The soil extractants, the chemical solution used to pull nutrients out of the soil so we can measure nutrients, is just as important as the measurement itself. When we talk about soil testing, we often focus on the numbers: phosphorus at 12 ppm, potassium at 180 ppm, base saturation at 75 percent. But what’s often overlooked is how those numbers were generated in the first place.
At Ward Laboratories, we use extractants that are regionally appropriate, scientifically validated, and calibrated to crop response.
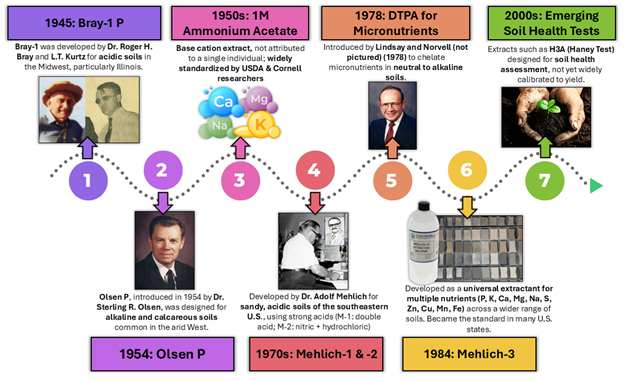
Calibration involves field trials where the extract test values are statistically correlated to plant uptake or yield response across soil types, crops, and conditions (Figure 1). Without this, the soil test number is just a relative value.
A Short History of Soil Extractants
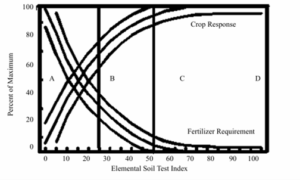
Soil testing in the U.S. gained traction in the 1940s and 50s as universities developed extractants suited to their regional soils, often led by pioneering soil scientists:
- Bray-1 was developed by Roger H. Bray and L.T. Kurtz in 1945 for acidic soils in the Midwest, particularly Illinois. It uses a dilute acid and fluoride to extract available phosphorus.
- Olsen P, introduced in 1954 by Sterling R. Olsen, was designed for alkaline and calcareous soils common in the arid West. It uses sodium bicarbonate to avoid dissolving calcium carbonate.
- Mehlich-1, -2, and -3 were created by Adolf Mehlich, with Mehlich-3 introduced in 1984 as a multi-nutrient extractant effective across a wider range of soil types. It was originally adopted in the Southeast.
- DTPA (diethylenetriaminepentaacetic acid) was introduced by Lindsay and Norvell (1978) to chelate micronutrients in neutral to alkaline soils, improving availability estimates for Zn, Fe, Mn, and Cu.
- Ammonium acetate (1M, pH 7) became the standard for determining exchangeable cations and CEC. Though not tied to one individual, it gained widespread adoption based on early work by soil chemists at Cornell and USDA labs.
Figure 2 shows a timeline for the development of these tests.
Each method was developed for a specific purpose and soil chemistry, and using the wrong one can misrepresent what’s truly available to plants in your soil.
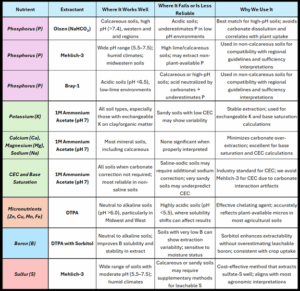
Which Extractants Work Where, and Why Some Fail
Olsen P (0.5 M sodium bicarbonate, pH 8.5)
This bicarbonate-based extractant works well in high-pH and calcareous soils because it avoids dissolving calcium carbonates. Studies in Idaho and Nebraska show that Olsen P correlates well with crop response in western soils.
Bray-1 P
Bray-1 is acidic, and in the presence of free lime or carbonate, that acid is neutralized. The result? A falsely low phosphorus reading; Bray-1 underestimates P when CaCO₃ is present, often by 50 percent or more.
Mehlich-3
Mehlich-3 is a strong, buffered acid extractant that works well for P, K, and some micronutrients. It performs reliably in a wide range of soils, but with some caveats:
- In high-pH calcareous soils, it can overestimate Ca and Mg due to partial carbonate dissolution.
- Mehlich-3 can artificially inflate CEC in soils with pH >7.3.)
- Still, it’s one of the most commonly used extractants in the U.S. and performs well when interpreted properly.
Is Mehlich-3 Calibrated for All Elements?
The short answer: No.
Mehlich-3 is not universally calibrated for all elements in all regions and soil types.
While it’s very effective at extracting many elements, that doesn’t mean each of those extractions has a reliable calibration curve to actual crop response. The main elements that Mehlich-3 is calibrated for are P, S, and sometimes Calcium (Ca), Magnesium (Mg), and Sulfur (S), if they’re reported, but they’re often less frequently tied to crop yield.
Even with this understanding, many growers still use this data not knowing they may be under- or oversupplying these key nutrients.
Below is a summary table of the various extracts, where they work, where they don’t, and when to use them.
Lab Fertility Recommendations: Same Results, Different Interpretations
Why do two labs using the same extractant give different fertilizer recommendations? Because the interpretation philosophies differ:
- Apply nutrients only when levels are low.
- Apply more than the crop needs to raise and sustain soil levels.
- Combine both depending on nutrient and crop.
Some instances have shown that a soil with 20 ppm Mehlich-3 P, for example, could result in fertilizer recommendations ranging from 0 to 90 pounds of P₂O₅ per acre, depending on the state. This reinforces the need for local calibration and interpretation, which Ward Labs applies using Midwest-based correlations and research trials (University of Lincoln-Nebraska & South Dakota State University).
It’s About Fitness for Purpose
Extractants aren’t just lab reagents; they’re decision-making tools. And like any tool, they have to fit the job. At Ward Laboratories, we don’t just use extractants because they’re standard. We use them because they’ve been tested, validated, and proven to work in the soils our clients manage.
Whether you’re farming in the Great Plains, irrigating in western Nebraska, or managing pasture in the Sandhills, your soil’s chemistry deserves the right extractant and the right interpretation.




1 Comment-
Thank you. Nice blog of explanation.