When it comes to hay testing, producers commonly grumble about the variation in Relative Feed Value (RFV) and protein content, based on their observations and what the lab reported.
Producers often have these concerns, because the RFV determines the price of a forage and how much customers are willing to pay. An underestimated RFV can result in decreased profit for forage producers. A couple weeks ago, I attended the NIRS Consortium annual meeting where Rocky Lemus gave a very informative talk about the importance of proper sampling, which addressed the producer concerns I often hear. Variation in forage test results can come from in the field, storage, sampling, and in the lab. Here are 14 common sources of variation within a forage sample:
- The leaf : stem ratio, forages with a higher leaf : stem ratio are typically higher in protein and RFV. This is because the leafy portion of the plant contains more protein than the stems. Additionally, the stems are a structural part of the plant containing higher amounts of fiber. Acid detergent fiber (ADF) and neutral detergent fiber (NDF) are used to calculate RFV therefore, forages and hays with more leaves and less stems are lower in ADF and NDF and higher in RFV.
- The weed content of a forage can affect the RFV. Weeds are high fiber plants, so the more weeds contaminating the forage or hay the lower the RFV. Weeds also tend to be lower in protein, thereby also affecting the nutritional content of the forage.
- Baling conditions can also affect the RFV of a hay. If hay is baled under moist conditions or after having been rained on, the water-soluble sugars have been removed from and plant. The percent of ADF and NDF are increased due to the absence of sugars. The result is a RFV and lower energy forage.
- Species of forage also affects RFV. This is not news to hay producers or livestock feeders. It is well understood that legume forages such as alfalfa and clover are typically higher protein and higher RFV, than grasses. This is a result of the leaf : stem ratios.
- The maturity of a plant can also affect the feed value. Older more mature plants are more fibrous, and they typically have a lower RFV than a lush growing forage.
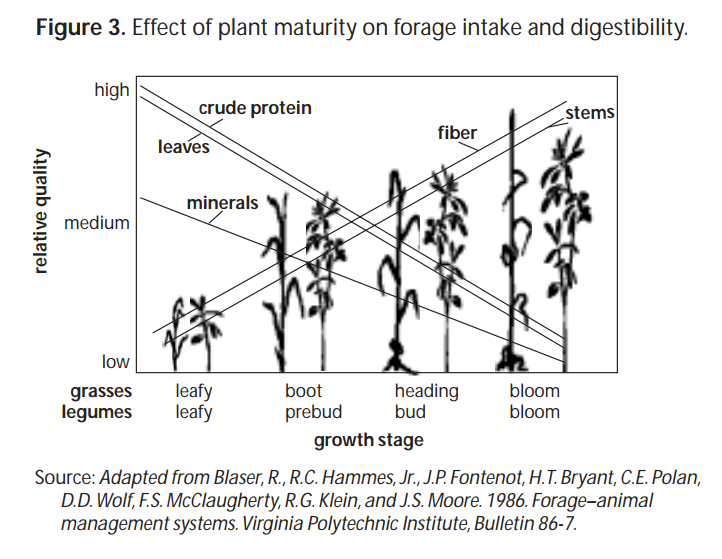
- Fertilization can result in higher protein in a forage and lower ADF and NDF. Fertilization management may help produce high quality forages. Be cautious to avoid creating a high nitrate forage by applying too much fertilizer.
- Proper storage of a baled hay is very important. Reduce ground contact as this will result in accumulation of moisture from the ground and a decreased RFV. Protect your baled hay from the elements to avoid losses of soluble sugars and protein. Wind and rain alike can remove the leafy portion of the plant thereby decreasing protein and RFV.
- Division of forages into separate lots can affect the accuracy and representation of a forage sample. Lots should be defined by both species and field from which it was baled. For example: If there are three fields two alfalfa and one grass, the lots need to be separated by not only species, but also field as one location may have a differing quality that the other based on management, precipitation differences, or topographical differences. Therefore, I would send three separate hay samples to Ward Laboratories, INC. for NIR testing.
- Proper sampling procedure is very important. Using a hay probe is the key to ensuring a representative sample. Hay probes can accurately represent the leaf : stem ratio, whereas using a hand grab can result in the leafy portions falling through fingers and obtaining an overrepresentation of stems and a lower RFV. Additionally, with a hand grab only one layer of the bale can be grabbed and to ensure a representative sample it is important to sample several inches inside the bale. For more guidelines and to become a certified hay tester visit the National Forage Testing Association.
- The number of cores taken is another source of variation when testing. The recommendation is to combine 20 randomly selected cores. The difference between taking 20 cores and 10 cores can cause variation in crude protein by up to 5%, meaning taking only 10 cores could either over estimate nutrient values or under estimate them.
- It is very important to ensure proper treatment of samples in delivery. If the sample is hay, it is typically dry enough to not have cause for concern. If it is a fresh forage clipping, check the moisture, if it is very damp rotting can occur on its way to the lab in just a few days stuffed in a box with other samples or envelopes. Of course, the portion of the plant that rots first is the leaves, so the RFV decreases when this happens on the way to the lab.
- Splitting in the lab can also affect RFV. Ward Laboratories, INC. uses the cone and quarter method on all forage samples that come in the lab. It is very important that when the lab splits the sample for the portion to be tested it represents the sub sample given to us. Sometimes, it is requested to send the sample on to other labs, when this happens, the sample is split into two – three sub samples and the NIR scans are checked to ensure the sub samples nutritional values repeat. This way we can keep some sample in our lab in case further testing is requested and it is a good way to check that our sub-sampling procedure is accurate.

- Drying of the sample in the lab can result in heat damage to the sample therefore again decreasing RFV. Ward Laboratories, INC dries samples in an oven at 64°C before grinding, the typical dry matter after grinding is between 95-97% dry matter. Other labs use microwave ovens for faster drying time, however using a microwave does result in higher incidence of heat damage to samples.
- Grinding of the sample can also make a difference as to how it scans NIR. Ward Laboratories, Inc. grinds samples through a 1mm screen. More coarse grinding can cause inaccurate results on the NIR instrument.
When questioning results of a forage or hay sample, consider all the sources of variation that went into that sample. Sampling plants is tricky business as it is a variable material. Always do your best to take a representative sample. Call the lab if you have questions or concerns before taking your sample or interpreting your results.




[…] Precise ration and diet formulations improve profitability by reducing the occurrence of over or under supplementation to reach animal production goals. To produce a precise and accurate ration or diet, forage testing must be done, otherwise producers are just guessing about the nutrient content of the forage. Forages are variable plant material. As the feed and NIR reviewer at Ward Laboratories Inc., I have seen alfalfa hays and grasses vary from a crude protein level of about 15% to 25% and 4% to 18% on a dry basis respectively. The fiber content of various forages is also variable. Acid detergent fiber (ADF) is used to calculate the total digestible nutrients (TDN) of the feed, so variation in ADF affects energy supplementation. Neutral detergent fiber (NDF) affects how much of a forage or hay the animal will consume. Minerals are also variable in forages and obtaining an idea of the mineral content may also affect mineral supplementation strategies, such as which mineral to feed or possibly creating a custom mineral mix. If you would like to learn more about forage variability read Quarrels About Quality: 14 Sources of Variation in Forage and Hay Testing. […]
[…] Precise ration and diet formulations improve profitability by reducing the occurrence of over or under supplementation to reach animal production goals. To produce a precise and accurate ration or diet, forage testing must be done, otherwise producers are just guessing about the nutrient content of the forage. Forages are variable plant material. As the feed and NIR reviewer at Ward Laboratories Inc., I have seen alfalfa hays and grasses vary from a crude protein level of about 15% to 25% and 4% to 18% on a dry basis respectively. The fiber content of various forages is also variable. Acid detergent fiber (ADF) is used to calculate the total digestible nutrients (TDN) of the feed, so variation in ADF affects energy supplementation. Neutral detergent fiber (NDF) affects how much of a forage or hay the animal will consume. Minerals are also variable in forages and obtaining an idea of the mineral content may also affect mineral supplementation strategies, such as which mineral to feed or possibly creating a custom mineral mix. If you would like to learn more about forage variability read Quarrels About Quality: 14 Sources of Variation in Forage and Hay Testing. […]